 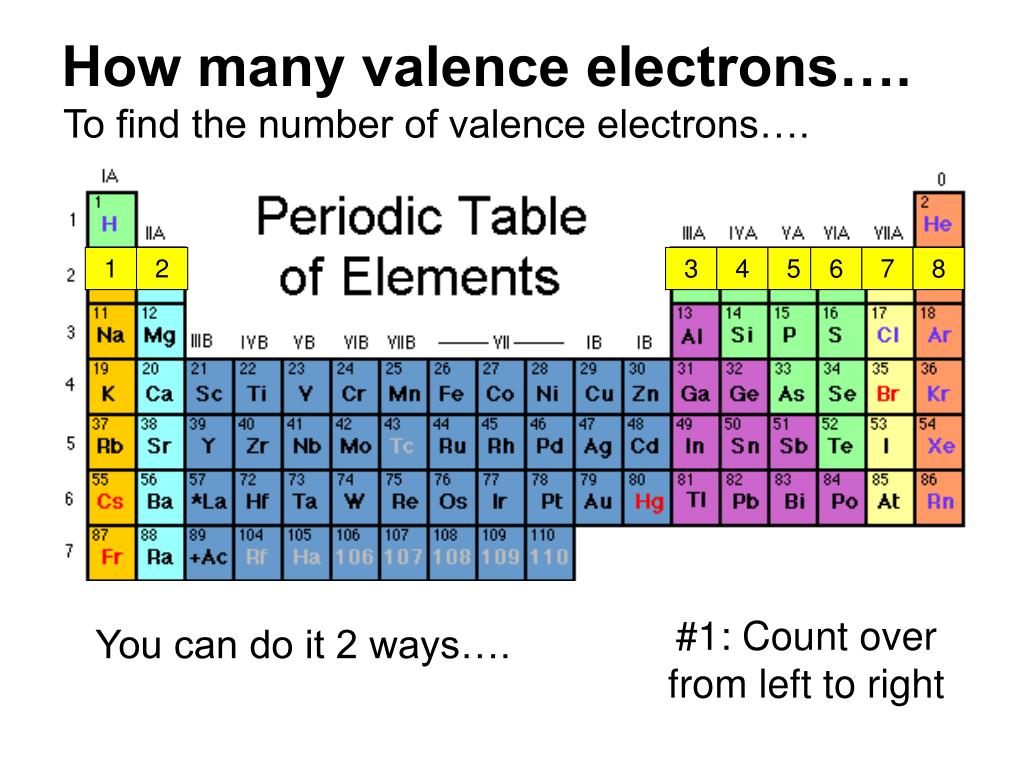
In practice, chemists simplify the notation by using a bracketed noble gas symbol to represent the configuration of the noble gas from the preceding row because all the orbitals in a noble gas are filled. When we reach neon, with Z = 10, we have filled the 2 p subshell, giving a 1 s 22 s 22 p 6 electron configuration:Īs we continue through the periodic table in this way, writing the electron configurations of larger and larger atoms, it becomes tedious to keep copying the configurations of the filled inner subshells. So 1s2, 2s2, 2p2 is the electron configuration for carbon. And then we have, we're in the 2p1 and then 2p2. Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons. If I wanted to write an electron configuration for carbon, well it would be 1s2. Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons. Similarly, fluorine has the electron configuration 1 s 22 s 22 p 5: Fluorine has one electron pair in the 1 s orbital, one electron pair in the 2 s orbital, and 2 electrons pairs with one unpaired electron in the 2 p orbital. Another common method of categorization recognizes nine element families: Alkali Metals: Group 1 (IA) - 1 valence electron. The number of valence electron remains constant even when the number of shells expands as we move from the top to the bottom of a group. These electrons determine the way in which the atom will combine with other. Here, we just look up the element in the periodic table to get its specific location. In chemistry, those electrons found in orbits farthest from the nucleus of the atom. For example, a hydrogen atom with one unaired dot can make one bond as. Generally, each unpaired dot can make one bond. The electron dots in the Lewis structure are a convenient way to determine how many bonds an atom of an element can make. Oxygen has one electron pair in the 1 s orbital, one electron pair in the 2 s pair, and one electron pair, and 2 unpaired electrons in the 2 p orbitals. Using the Periodic Table In order to count the valence electron of an element, this is the most used technique. 1: Lewis symbols or electron-dot symbols of the first twenty elements in the periodic table. Atoms in Groups 13 and 18 have 3 and 8 valence electrons. 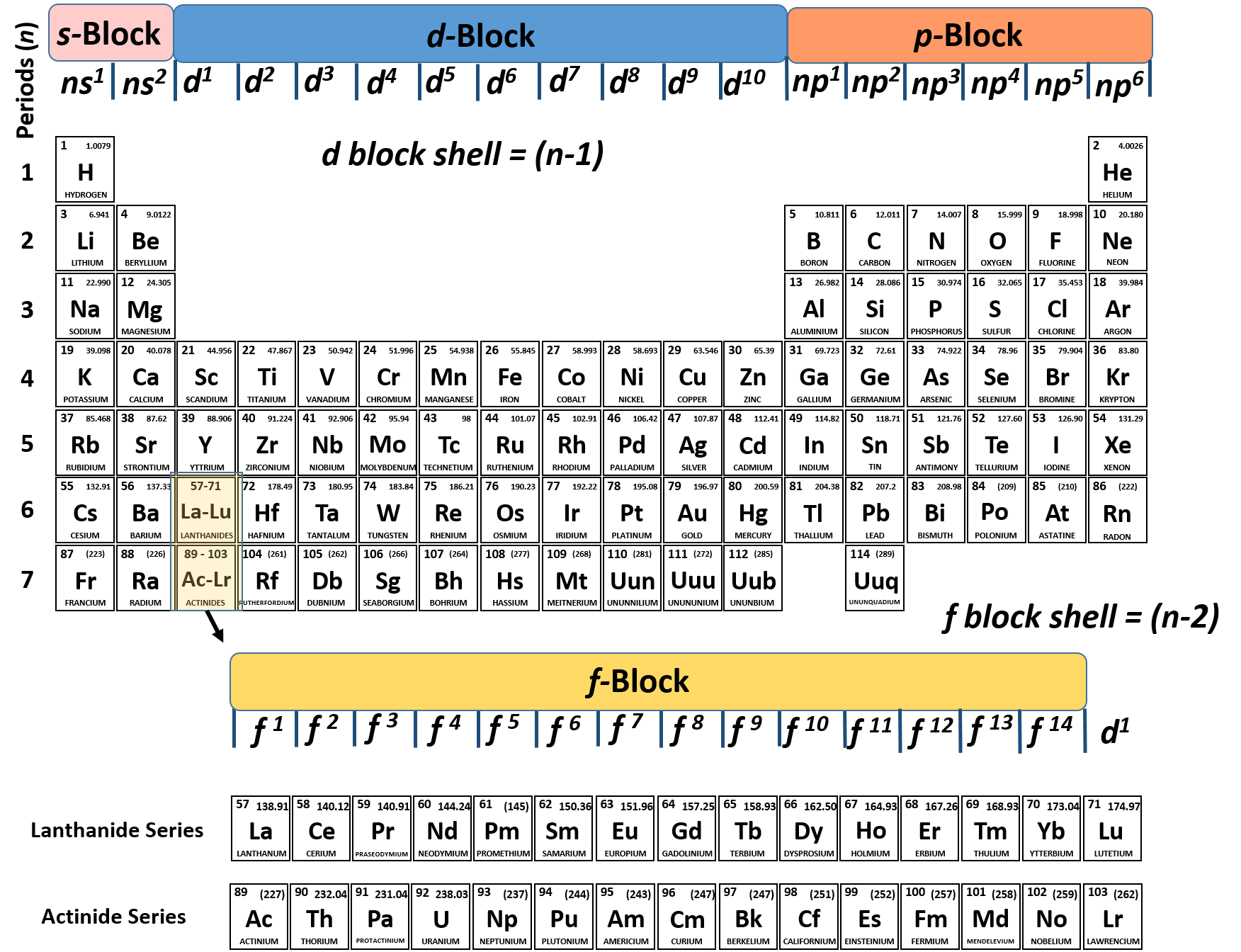
For example, atoms in Groups 1 and 2 have 1 and 2 valence electrons, respectively. You can easily determine the number of valence electrons an atom can have by looking at its Group in the periodic table. Because all the 2 p orbitals are degenerate, it doesn’t matter which one has the pair of electrons. Valence electrons are the electrons present in the outermost shell of an atom. One electron must be paired with another in one of the 2 p orbitals, which gives us two unpaired electrons and a 1 s 22 s 22 p 4 electron configuration. Electron affinity increases upward for the groups and from left to right across periods of a periodic table because the electrons added to energy levels become closer to the nucleus, thus a stronger attraction between the nucleus and its electrons. The electron configuration of nitrogen is thus 1 s 22 s 22 p 3.Īt oxygen, with Z = 8 and eight electrons, we have no choice. In simpler terms, it can be defined as something similar to the radius of a circle, where the center of the circle is the nucleus and the outer edge of the circle is the outermost orbital of electron. For example, the electron dot diagram for iron (valence shell configuration 4s 2 3d 6) is as follows. Atomic radius is generally stated as being the total distance from an atom’s nucleus to the outermost orbital of electron. The electron configuration of nitrogen is thus 1 s 22 s 22 p 3.Īt oxygen, with Z = 8 and eight electrons, we have no choice.\): Copy and Paste Caption here. For atoms with partially filled d or f subshells, these electrons are typically omitted from Lewis electron dot diagrams. Valence electrons are those outermost electrons that dictate the properties of the atom and are involved in chemical bonding. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
